4,49 €
Mehr erfahren.
- Herausgeber: epubli
- Kategorie: Wissenschaft und neue Technologien
- Sprache: Deutsch
Are the chemical properties of the elements, instead of an electron system, better explainable by a proton-neutron system of the atom? Already Mendelejew and Meyeer saw the atomic mass as causal! Until today there is a contradiction between the views of Dmitri Mendeleev and Lothar Meyer and those of Niels Bohr on the cause of the chemical properties of the elements, which has never been discussed. While Bohr and with him today's science assume that the chemical properties of the elements depend on almost massless electrons of the atomic shell, Dmitri Mendeleev and Lothar Meyer saw it quite differently. In contrast, they postulated that the chemical properties depend on the atomic mass. However, as we know today, the carriers of the atomic mass are not the electrons, but the protons and neutrons. Therefore, the question arises whether there is a proton-neutron system instead of the electron system on which the chemical properties and valences of the elements depend?
Das E-Book können Sie in Legimi-Apps oder einer beliebigen App lesen, die das folgende Format unterstützen:
Seitenzahl: 28
Veröffentlichungsjahr: 2021
Ähnliche
Is the Electron System a Misinterpretation of Bohr?
Helmut Albert
Imprint
Texts and Illustrations: © 2021 Copyright by Helmut Albert
Cover: © 2021 Copyright by Helmut Albert
Translation from German: DeepL plus
„Ist das Elektronensystem eine FehlinterpretationBohrs?“ (2021)
Responsible for the content:
Helmut Albert, Talstraße 63', 79102 Freiburg / Germany
hm.albert@t-online.de
E-Book: epubli - a service of Neopubli GmbH, Berlin
Inhalt
1.Atomic Structure and Atomic Mass
2.State of Science
3.Mendeleev's and Meyer's Theses
4.Bohr´s Atomic Theory
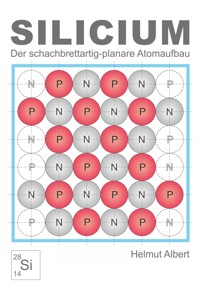
5.The Checkerboard-like Atomic Structure
6.Bibliography
7.List of Figures
8.Biography
Atomic Structure and Atomic Mass
Until today there is a contradiction between the views of Dmitri Mendeleev (1834-1907) and Lothar Meyer (1830-1895) versus those of Niels Bohr (1885-1962), on the cause of the chemical properties of the elements, which has never been addressed. While Bohr and with him the present science, assumed that the chemical properties of the elements depend on almost massless electrons of the atomic shell, Dmitri Mendeleev and Lothar Meyer saw it quite differently. In contrast, they postulated that the chemical properties depend on the atomic mass. However, as we know today, the carriers of the atomic mass are not the electrons, but the protons and neutrons. Therefore the question arises whether there is a proton-neutron system instead of the electron system, on which the chemical properties and valences of the elements depend?
At the beginning of the paper, the state of science concerning the periodic table and the atomic structure is presented. Thereby the shell model, or orbital model and the Bohr atomic model are shown as the atomic models which are decisive for today's atomic conception. Likewise, the nuclear shell theory, which was only published in 1948, is presented, which refers exclusively to the atomic nucleus. An important aspect of the atomic nucleus and its nucleons is the atomic mass, which is also examined in this chapter.
This is followed by a discussion of the content and significance of the periodic table of the elements. It is shown that the periodic table of the elements is not only a clear table of the elements, but can be understood as a starting point for a logically comprehensible atomic structure. The focus is also on the theses of Mendeleev and Meyer that the chemical properties of the elements depend on the atomic mass. Thereby the question arises, how their statements and theses are to be interpreted with today's knowledge about the basic building blocks of the so-called atomic nucleus?
The following chapter deals with the opinion of Niels Bohr. He represented, with regard to the chemical properties of the elements, a virtually opposite view as Mendelejew and Meyer. Niels Bohr, who based his Bohr atomic model of 1913 on the nuclear-shell atomic model of his teacher Rutherford, saw the chemical properties of the atoms as depending on the massless electrons in the atomic shell. He substantiated this view with his "Aufbauprinzip" published in 1921, in which he juxtaposed the protons with one electron per period in the circular arrangements of his electron system. Thus he could explain schematically electric charges of atoms.
Tausende von E-Books und Hörbücher
Ihre Zahl wächst ständig und Sie haben eine Fixpreisgarantie.
Sie haben über uns geschrieben: